From hydrogen and oxygen to fuel cell
The first fuel cells were invented in 1838 but they were not used commercially until the NASA space program developed a program using fuel cells to generate power for satellites and space capsules. Discover in this article how the MASS-VIEW® can be used to create a fuel cell.
Working principle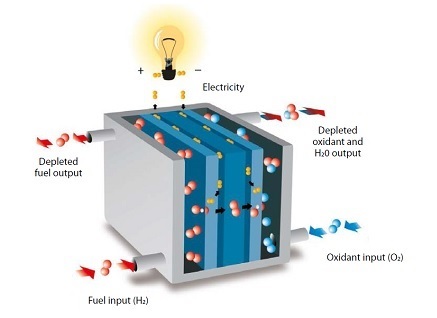 “Fuel cells convert chemical energy into electricity through an reaction of (usually) hydrogen and oxygen.“
“Fuel cells convert chemical energy into electricity through an reaction of (usually) hydrogen and oxygen.“
In contrast to a battery, where a reaction of chemicals present in the battery occurs, fuel cells need a continuous source of fuel and oxygen to produce energy.
As displayed in the picture, a fuel cells consists of an anode, a cathode and an electrolyte in which positively charged hydrogen move between the two sides of the cell.
- Anode: The starting point. Here, hydrogen molecules split into protons and electrons. Protons eagerly traverse to the cathode, while electrons embark on an external journey, creating direct current electricity.
- Cathode: The receiver. Protons from the anode meet electrons from the external circuit. Together, they engage in a cosmic tango, forming water (H₂O) as their final act.
- Electrolyte: The bridge. Positioned between the anode and cathode, the electrolyte allows protons to shimmy across, completing the circuit.
An oxidation process commences in the anode that generates protons and electrons.
The protons that were developed flow from the anode to the cathode.
Direct current electricity is produced through the process of protons flowing from the anode to the cathode through the electrolyte and the electron moving from the anode to the cathode through an external circuit.
In the cathode a catalyst causes hydrogen, oxygen, and electrons to react to form water.
The journey from hydrogen and oxygen to a functional fuel cell involves intricate science and engineering.
The MASS-VIEW® plays a crucial role in monitoring and optimizing these processes, ensuring efficient energy production. 🚀🔬🔋
What we offer
Mass Flow ONLINE offers MASS-VIEW Meters, Controllers and Regulators that can assist in this production of cables and wires. To be ensured of a high quality product the production process has to be monitored closely and materials have to be measured precisely. The MASS-VIEW allows for direct measurements, regulation and control over the process with a high accuracy and rangeability that is clearly displayed on its OLED display.
- Special designed MASS-VIEW models that is suited for H2 (Hydrogen) and He (Helium).
- MASS-VIEW meters / regulators / controllers are available in wide flow ranges from 10 – 200 mln/min up to 1 – 500 ln/min.
- The compactness of the MASS-VIEW® allows to construct a portable and easy-to-use rig.
- Possibility to control a variety of gases, like Air, N2, O2, CO2, CO, N2O, Ar, CH4, C3H8 and C4H10.